|
The final gel formulation was also characterized rheologically. The microparticles were characterized via scanning electron microscopy analyses, Fourier transform infrared spectroscopy (FTIR) analyses, via determination of encapsulation efficiency, and via thermal analyses (both TGA and DSC). With the goal of utilizing the microparticles containing encapsulated NTZ in pharmaceutical formulations, one prepared microparticles containing NTZ via ionotropic gelation of sodium alginate. Microencapsulation is a process utilized in the incorporation of active ingredients within polymers aiming at, among other objectives, the prolonged release of pharmaceutical compounds and protection from atmospheric agents (viz. Nitrofurazone (NTZ) is usually employed in the topical treatment of infected wounds and lesions of both skin and mucosa. In addition, this sensor exhibits high selectivity, reproducibility, stability, and also was successfully used to directly determine nitrofurazone in the commerical antibacterial lotion with comparative sensitivity to high-performance liquid chromatography (HPLC), showing its promising application prospects. The DPV determination of nitrofurazone indicates that the linear range and detection limit of nitrofurazone are 1 − 50 µmol dm⁻³ and 0.25 µmol dm⁻³, respectively. 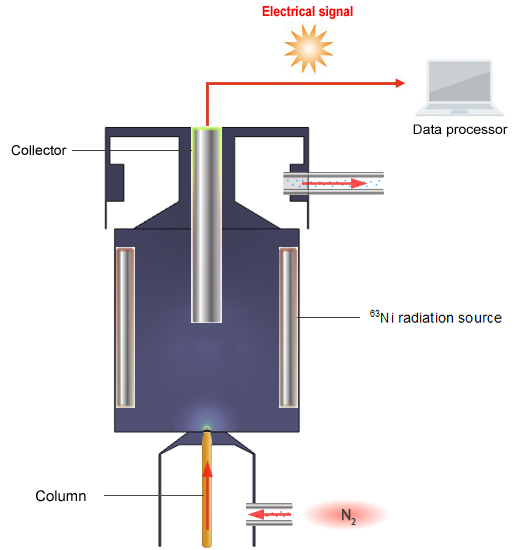
In pH 7.0 Britton-Robinson buffer solution, the as-prepared polyfurfural-electrochemically reduced graphene oxide modified glassy carbon electrode shows excellent electrocatalytic performance for the electrochemical reduction of nitrofurazone, and the reduction peak current is about 9.45, 1.31 and 1.25 times higher than that of the bare glassy carbon electrode, polyfurfural modified glassy carbon electrode, and electrochemically reduced graphene oxide modified glassy carbon electrode, respectively. The morphologies and properties of the sensor were characterized by electrochemical impedance spectroscopy (EIS), scanning electron microscopy (SEM), cyclic voltammetry (CV), and differential pulse voltammetry (DPV). 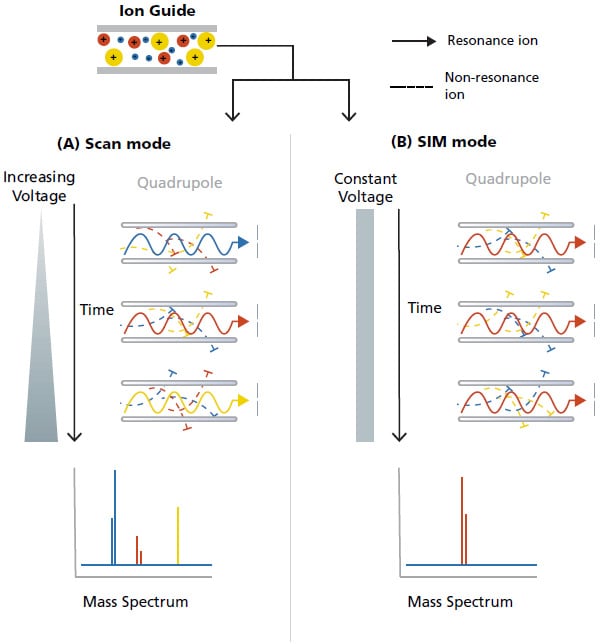
The mechanism of O-AgNPs interaction with drug was followed by DLS suggest that NFZ induces a time dependent nanoparticles aggregation.Īn electrochemical sensor based on a polyfurfural-electrochemically reduced graphene oxide modified glassy carbon electrode has been developed for the sensitive and rapid determination of nitrofurazone. The O-AgNPs showed high selectivity towards nitrofurazone (NFZ) with the lowest detection limit of 1.88 μM as was monitored by UV–vis spectroscopy. Moreover, chemosensing properties of the O-AgNPs were also tested against different drugs. Despite potent antimicrobial, anti-biofilm and biofilm eradicating activities, O-AgNPs were found to be non-toxic on cell lines. Stability of O-AgNPs studied by varying storage period, temperature, salt and pH of the medium were found quite stable probably because of oil suspension nature. The morphology of O-AgNPs were found spherical in nature, while the size of O-AgNPs ranges between 35 to 65 nm as established by AFM and DLS studies, respectively. FTIR analysis revealed that the carbonyl (CO) groups of Olive oil plays the most vital role in reduction of Ag⁺ and the nanoparticles synthesis. The chrome yellow color solution of O-AgNPs show the typical absorption maximum at 430 nm. Silver nanoparticles were synthesized using Olive oil (O-AgNPs) as reducing as well as capping agent and extensively characterized by UV–vis spectroscopy, fourier transform infrared, energy disperse spectroscopy, dynamic light scattering and atomic force microscopy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
